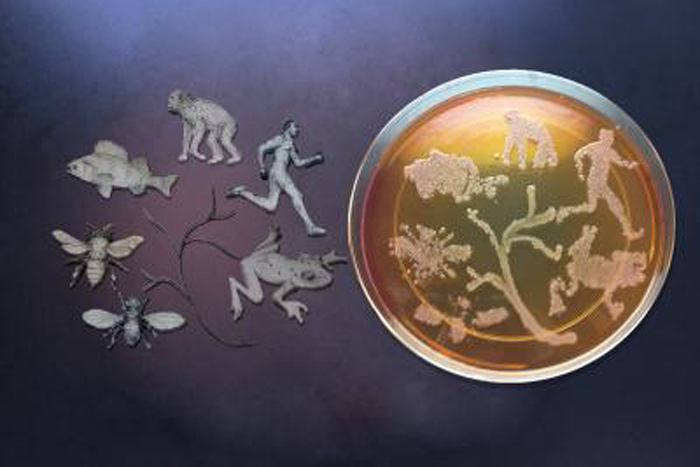
Evolution Influenced By Temporary Microbes
Life on Earth often depends on symbiotic relationships between microbes and other forms of life. A new theory suggests that researchers should consider how symbiotic microbes can influence the evolution of life on Earth, and possibly beyond.
Research exploring this new theory could one day lead to microbes that can help astronauts adapt to zero gravity and other environments, scientists added.
Recent findings suggest that the average human gut is home to roughly as many bacteria as there are cells in the human body — about 39 trillion microbes compared to 30 trillion human cells. This consortium of bacteria, altogether known as the microbiome, mostly consist of either harmless or friendly microbes. One well-known example is the bacterium E. coli, of which some strains help synthesize a critical vitamin, K2. A host and all its microbes can be thought of as a single entity known as a holobiont. [Packing Lunches for Space: Scientists Talk Astronaut Health on 1 Year Mission]
Article continues belowEvolutionary biologists often think about evolution as working on the level of organisms, with the individuals that are best adapted to their environments most likely surviving to pass on their adaptations to their descendants and perhaps to their species as a whole. A recent tweak to this concept is the so-called hologenome theory of evolution, which considers evolution working on the level of the holobiont.
However, some microbes do not fit well inside the hologenome theory of evolution for a number of reasons. They may not pass reliably from generation to generation, or they may only have a temporary influence on their hosts, said study lead author Jameson Voss, a preventive medicine physician at the United States Air Force School of Aerospace Medicine at Wright Patterson Air Force Base in Ohio. For instance, while the majority of the gut microbes in humans are stable day to day, only 60 percent of the strains there last beyond five years.
Now, Voss and his colleagues suggest that evolution may also be influenced by what they call "pawnobes" — microbes that are not stable, permanent residents of a holobiont, but which a host can still purposefully manage for its own benefit.
Pawnobes are members of a holobiont, but they are not wholly dependent on their hosts. The fact that pawnobes do not strictly depend on the survival of their hosts makes them more free to evolve independently and more rapidly than their hosts.
The potential that pawnobes have for genetic adaptability means that their hosts could benefit from new adaptations of the pawnobes within a single generation of the hosts. Such innovation could greatly speed the evolution of holobionts.
The researchers chose the word "pawn" for its numerous connotations. For example, in the game of chess, pawns are not critical pieces, but they remain strategically important, and if they survive and advance to the end of the board, they can be upgraded to a more functional piece. Another set of analogies can be drawn from pawn stores, which exist at the border between regulated exchanges and unregulated barter.
Just as purposefully selecting animals and plants thousands of years ago helped humans go beyond hunting and gathering to trigger the agricultural revolution, perhaps intentionally domesticating microbes could help trigger a similarly important shift, Voss and his colleagues suggest. Increasingly people are consuming probiotics — microbes that are believed to provide health benefits when ingested — and future research could purposefully engineer probiotics to account for complex interactions between different members of the pawnobiome.
To test this "pawnobiome theory of evolution," the researchers suggest experiments to see how much the pawnobiome can change over relatively short time scales, and how much effect such modifications might have on their hosts. For instance, mice naturally eat feces, which are loaded with gut microbes, so that scientists could take stool from the leanest and otherwise healthiest mice and feed it to other mice with unhealthy diets to see which rodents benefit most, and promising microbes could lead to further experiments in animals and finally humans.
"Costs and regulatory issues may be obstacles that may present in future research or development in this area of study," Voss said. "However, conducting initial studies using insect models may help overcome some of these hurdles until our understanding in the area of microbial artificial selection matures."
Further research into the pawnobiome could also benefit astrobiology and exobiology, Voss said. For instance, pawnobiome experiments "could be tested for adaptation to zero gravity or other space-related environments," Voss said.
The scientists detailed their findings in the August issue of the journal Frontiers in Microbiology. This work was partly funded by theExobiology & Evolutionary Biology element of the NASA Astrobiology Program.
This story was provided by Astrobiology Magazine, a web-based publication sponsored by the NASA astrobiology program. Follow Space.com @Spacedotcom, Facebook and Google+.

Charles Q. Choi is a contributing writer for Space.com and Live Science. He covers all things human origins and astronomy as well as physics, animals and general science topics. Charles has a Master of Arts degree from the University of Missouri-Columbia, School of Journalism and a Bachelor of Arts degree from the University of South Florida. Charles has visited every continent on Earth, drinking rancid yak butter tea in Lhasa, snorkeling with sea lions in the Galapagos and even climbing an iceberg in Antarctica. Visit him at http://www.sciwriter.us