Radioactive decay: Discovery, process and causes
What is radioactive decay and is it possible to predict?

Breaking space news, the latest updates on rocket launches, skywatching events and more!
You are now subscribed
Your newsletter sign-up was successful
Want to add more newsletters?
Radioactive decay is the strange and almost mystical ability for one element to naturally and spontaneously transmute into another. In the process, those elements tend to emit deadly forms of radiation in the electromagnetic spectrum.
Radioactive decay is most commonly associated with nuclear disasters, such as at Chernobyl, where the resulting harmful radioactive decay in the surrounding area necessitated the creation of an exclusion zone. Radioactive decay occurs in atoms that become unbalanced, called radionuclides, according to the U.S. Environmental Protection Agency. These radionuclides follow their own unique path of decay, transforming into different elements until they reach a stable state.
The discovery of radioactivity
In the late 1800s scientists discovered X-rays, a high-energy form of radiation, and were wondering if other kinds of "rays" were bouncing around. Simultaneously, scientists were just beginning to explore the nature of phosphorescence, the ability of some materials to glow after being exposed to X-rays.
Article continues belowFrench scientist Henri Becquerel was experimenting with various phosphorescent materials, including uranium. He soon discovered that all phosphorescent materials failed to leave an image on a photographic plate if he covered the plate, but uranium still did. That meant that uranium was generating its own kind of radiation that was different from phosphorescence. In the naming fashion of the time, this kind of radiation became known as Becquerel rays.
Related: The quest to find one of the most elusive particle decays in the universe
Other scientists picked up the trail from there, discovering that uranium really was quite odd. In the process of emitting radiation, it transmuted into other elements — something nobody had ever seen before, according to NobelPrize.org. They found that other elements were sometimes capable of the same trick. Pierre and Marie Curie famously discovered radium and polonium and coined the term "radioactivity," which quickly won out over "Becquerel rays."

Types of radioactive decay
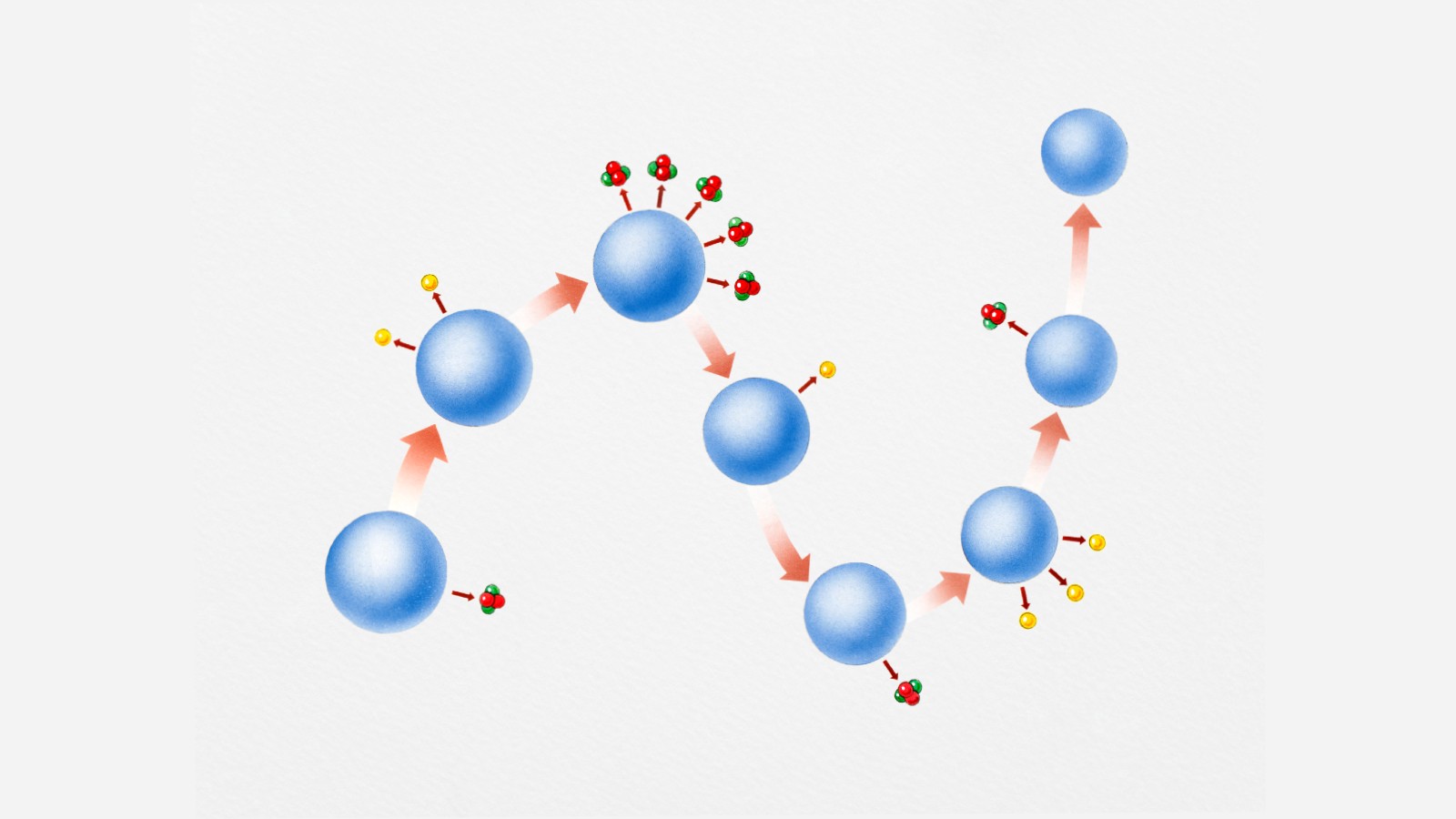
The Curies and other scientists found that radioactive elements emitted three kinds of radiation as they underwent this transmutation process.
Breaking space news, the latest updates on rocket launches, skywatching events and more!
Alpha radiation, can be blocked easily with nothing more than a sheet of paper.
Beta radiation, requires something heftier, like a metal sheet.
Gamma-rays, require an entire block of lead to be stopped, according to the United States Environmental Protection Agency.
What causes radioactive decay?
It wasn't until physicists had a quantum model of the atom that they were able to understand what was going on with radioactive decay. They found that the atomic nucleus was really a loose, gooey bag of protons and neutrons all sloshing around against each other. That gooey bag had a lot of complicated physics going on inside it.
For one, there's the strong nuclear force itself, which is responsible for forming triplets of quarks into neutrons and protons. Some of this strong force "leaks out" of the neutrons and protons, however, and it's that leftover force that is responsible for forming atomic nuclei.
Second, the protons in a nucleus are positively charged, and like-minded charges absolutely loathe being close to each other. The protons are constantly trying to push themselves away from each other, but they are usually kept in check by the gluing ability of the strong nuclear force, according to AtomicArchive.com. Occasionally, the weak nuclear force makes an appearance too, with its ability to transform a proton into a neutron and vice versa.
Most atomic nuclei are stable most of the time; the balance of forces inside them keep them together. But sometimes, things can become unbalanced.
If there are too many protons, for example, the electrostatic repulsion of those protons destabilizes the nucleus. Or, if there are too many neutrons, the strong force isn't always capable of keeping everything together and preventing neutrons from wandering away.
If an atomic nucleus isn't in its lowest-energy state possible, radioactive decay can happen when the nucleus reshuffles and reorganizes itself to find a new stable situation. The different ways a nucleus can rearrange itself lead to the different kinds of radiation.
Alpha radiation is actually an entire helium nucleus (two protons and two neutrons) splintered off larger elements.
Beta radiation is an electron ejected when a proton turns into a neutron.
Gamma radiation is the release of a high-energy photon, according to the United States Nuclear Regulatory Commission.
Is radioactive decay predictable?
Radioactive decay is totally random. If you were to take a single atom, you would never be able to predict exactly when that atom will decay. This is because even highly unstable nuclei are bound together. There's still a strong force "bag" holding all the sloshing protons and neutrons inside it.
The atom might prefer to have a different combination of protons and neutrons, but it has to get there somehow. The only way to get there is by removing parts of itself or changing one of its components, which costs a bit of energy.
The atom will be in a lower-energy state, but it has to spend a little to get there.
Is radioactive decay a chemical process?
In classical physics, radioactive decay could never happen, because it is not possible spend energy that does not exist. Quantum mechanics allows for this to happen, but it does so randomly. Every once in a while, a slightly unstable atomic nucleus will spontaneously decay into something else.
Even though we can never know when a single atom will decay, we can build statistics of how a whole population of atoms will decay. That's the idea behind a half-life.
For example, a single lump of uranium contains almost countless atoms. Every second that goes by, you are never sure which atom will decay, but you can feel pretty confident that some atom, somewhere in the lump, will do so. This means the half-life tells you how long it will take for half the mass of radioactive material to decay, according to HyperPhysics.
Additional resources
The book “Marie Curie and Radioactivity” is a great illustrated resource for younger readers to introduce them to the concept of radioactivity and the pioneering work of Marie Curie.
For older readers, try out "Radioactivity: A History of a Mysterious Science" by Marjorie C. Malley.
Steve Weatherall gives a great Ted-ED presentation about radioactivity in this video.
Learn more by listening to the "Ask a Spaceman" podcast, available on iTunes and askaspaceman.com. Ask your own question on Twitter using #AskASpaceman or by following Paul @PaulMattSutter and facebook.com/PaulMattSutter.
Bibliography
HyperPhysics
United States Nuclear Regulatory Commission
AtomicArchive.com
NobelPrize.org

Paul M. Sutter is a cosmologist at Johns Hopkins University, host of Ask a Spaceman, and author of How to Die in Space.

