Martian Samples Risk Earth Contamination in Search for Life

NASA's upcoming Mars 2020 rover is designed not only to hunt for signs of life on the Red Planet, but also to collect samples that might one day get returned to Earth. However, researchers say they face major challenges in ensuring that the six-wheeled robot does not contaminate these samples with molecules from Earth that might be mistaken for signs of Martian life.
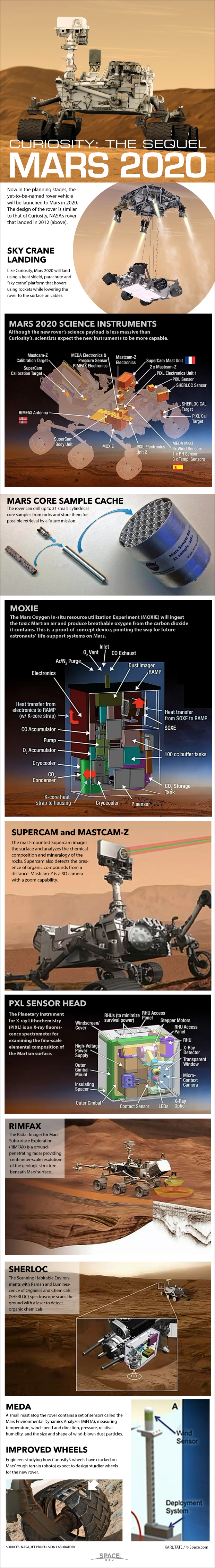
In many ways, the rover of the Mars 2020 mission — so named for its planned launch in 2020 — is built much like the Curiosity rover that landed on the Red Planet in 2012.
However, whereas Curiosity sought to find out how habitable Mars once was — that is, how capable it was of supporting life as it is known on Earth — "the motivating aspect of the Mars 2020 mission is astrobiology: to look for biosignatures, the signatures of life," according to Kenneth Farley, a project scientist for the Mars 2020 rover mission at the California Institute of Technology in Pasadena. [NASA's Mars 2020 Rover Mission in Pictures]
For instance, both Curiosity and Mars 2020 have instruments that can analyze what chemicals are in Martian rocks, including organic molecules that could hint at life. However, Mars 2020 can see if organic compounds are present in structures within rocks, which could provide stronger evidence of life, Farley told Space.com.
"When one looks at the oldest fossils on Earth, known as stromatolites, you can see spatial patterns when it comes to the organic molecules. For instance, banding consistent with algal mats, instead of a random distribution from a process that has nothing to do with life, such as interplanetary dust particles falling onto the planet," Farley said.
Unless Mars 2020 detects very obvious evidence of life, "high-confidence detection of life would require tools that are not presently flyable," Farley said. "This means samples with potential biosignatures would have to be brought back to terrestrial laboratories on Earth."
Keeping it clean
The Mars 2020 mission aims to collect up to 40 15-gram samples from the Red Planet. While the rover does not have the difficult task of returning these samples to Earth, just the act of collecting samples in a way that does not contaminate them is quite challenging, Farley said.
Breaking space news, the latest updates on rocket launches, skywatching events and more!
"The challenge for us is twofold," Farley said. "We have to prevent biologic contamination, and we have to prevent organic contamination. Meeting both rigid requirements simultaneously leads to complications. The thing that drives this complexity is the issue of organic contamination."
Organic molecules include most molecules containing carbon. Preventing organic contamination means, for instance, that samples cannot be placed in bags made of plastic, since plastics are organic polymers. "Organic polymers are great at keeping out microbial cells, but terrible in terms of organic contamination," Farley said.
"To give you an example of how challenging this task is, if you take a metal surface and you fire it — put it in a high-temperature oven with oxygen to burn off all the organic matter on it — and then place it in an ultra-clean lab, within minutes, it will accumulate organics from the atmosphere because its surface is extremely active," Farley said. "Bare metal surfaces are basically magnets for volatile organic materials."
To overcome this problem, the researchers started with titanium tubes whose inner surfaces were chemically modified to become titanium nitride. This material is much less active in terms of "glomming onto organics from the atmosphere," Farley said.
These sample-collecting tubes are then placed with their open sides down into a rack that is as clean as the scientists can get them. The gap between each tube and the walls of the rack is between 100 microns to a few millimeters wide, or so narrow "that it doesn't allow any appreciable flow of organic molecules," Farley said.
The rack of tubes is loaded into a robotic sampling and caching system on the rover about the size of a small microwave oven, Farley said. Although NASA will make this system as clean of contaminants as it can, "there will inevitably be problems," Farley said.
For instance, wires inside this system are likely covered in plastic, and these coverings can give off organic gases. "We will use minimal outgassing components, but once we seal the system, all that outgassing is going to be trapped inside the box," Farley said.
To deal with this problem, tubes leading into the system will pump in "an ultra-clean gas, probably nitrogen, constantly flushing accumulation of outgas products, probably right up to launch time," Farley said. "It's absolutely essential to keep this running."
At launch, as the rocket ascends, "the whole spacecraft will depressurize," Farley said. "In space, the nature of the way organic matter moves changes. It can no longer diffuse — it will basically stick on the walls instead, because of the cold and vacuum. So during the flight to Mars, we don't have to worry about contamination."
Mars2020 is using the same heat shield design as Curiosity, to protect it as it enters Mars' atmosphere. And, "it has the same rocket, and it's getting deployed by a sky crane just like Curiosity," said Farley.
"We don't invent a new rocket for each space mission; if we have the right tools, we reuse them," Farley said in a conversation with Space.com. "This reduces risk and money, and it frees up the engineering side to work on new things."
After the rover lands on Mars, the Red Planet's warmth and thin atmosphere make outgassing from components in the spacecraft a potential problem again. To address this problem, the rover will open a door so that any outgassed chemicals vent out to the Martian atmosphere, Farley said.
When samples are drilled from Mars, they are deposited into the tubes, which are then plugged up with a cap made mostly of titanium, Farley said. These tubes will then get deposited at a site where a future mission can pick them up. Bare metal tubes can reach about 212 degrees F (100 degrees C) in the sun, hot enough to destroy any organic samples within them. As such, NASA will cover these tubes with a reflective coating so that it will get no more than 18 F (10 C) hotter than their surroundings, Farley said.
Returning to Earth
Much remains unknown about what a future sample-return mission will look like. One possibility is that astronauts will retrieve these samples, while another is that a rover will, Farley said.
If a rover picks up the samples, there are two scenarios envisioned, Farley said. "You can have a rover with limited capabilities, one that does not do much if any science, that will drive to where we left the samples, pick them up, put it in the nose cone of a rocket on its back, and launch the rocket into orbit, and then a second mission captures this orbiting sample and brings it back to Earth," he said. "Another possibility is a fetch rover that goes to and from a base station where the rocket is."
The mission that returns the samples must take care not to potentially contaminate Earth. "We think it's incredibly unlikely that anything on Mars will be harmful to Earth, but we can't take the risk," Farley said. "We'll probably take the container that holds the samples and put it in another container that will be welded shut."
The hope is that the ancient rocks of Mars can yield insights on early Earth and, potentially, on the origins of life.
"If you look at Earth and ask how much rock we have that is older than 3.7 billion years old, the answer is almost none. On the other hand, Mars is freeze-framed in time, covered in rocks that are billions of years old, and so can tell us a lot about interesting things that happened early in the solar system," Farley said. "We know that Mars and Earth are different, but we believe that early Earth and early Mars were very similar, so by bringing samples back from Mars, we can look at environments that could shed light on the origin of life."
Follow Charles Q. Choi on Twitter @cqchoi. Follow us @Spacedotcom, Facebook and Google+. Original article on Space.com.

Charles Q. Choi is a contributing writer for Space.com and Live Science. He covers all things human origins and astronomy as well as physics, animals and general science topics. Charles has a Master of Arts degree from the University of Missouri-Columbia, School of Journalism and a Bachelor of Arts degree from the University of South Florida. Charles has visited every continent on Earth, drinking rancid yak butter tea in Lhasa, snorkeling with sea lions in the Galapagos and even climbing an iceberg in Antarctica. Visit him at http://www.sciwriter.us